|
Treatment with infliximab alone was continued with no relapse norĮvidence of disease recurrence. The orogenital ulceration and of the posterior uveitis. In a few weeks there was a marked improvement of Through intravenous infusion at weeks 0, 2, 6, and 8, then every 8 weeks. Therefore, infliximab was administred at a dose of 5 mg/kg, 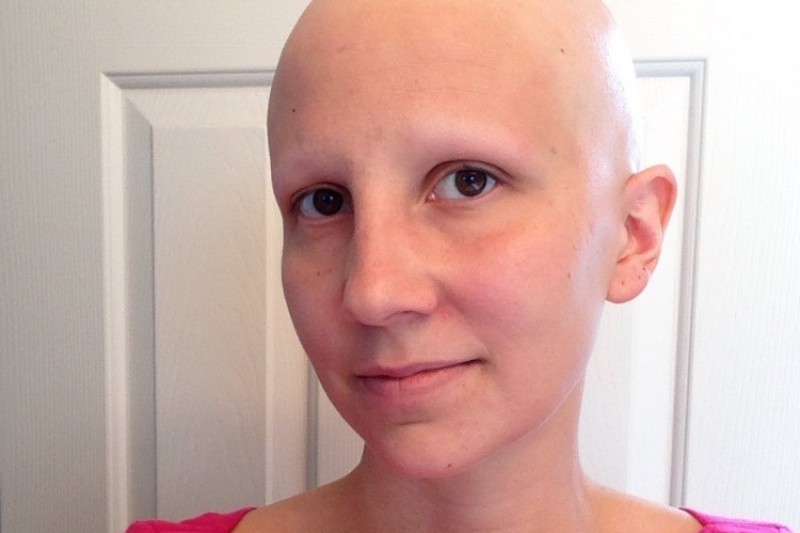
Unfortunately, worsening of the visual impairment was observed. Glucocorticoids and maintaining CSA, however, allowed a relapse of the disease and treatment with azathioprine was attempted. After 6 months, a relapse withĪctive eye disease was treated with corticosteroids and cyclosporine A (CSA), giving a rapid improvement. The orogenital ulcerationsĪnd the ocular manifestations have been successfully treated with prednisolone and colchicine. Ulcerations, severe uveitis, asymmetrical arthrithis involving knees, and HLA-B51 allele positivity. The patient is a 41-year-old man with Behçet disease (BD), whose diagnosis was performed on the basis of recurrent orogenital (c) A particular: bilateral eyelash alopecia. (a) Alopecia areata universalis within 6 months after beginning of the hair loss. We describe the first case of AA universalis that developed during infliximab treatment.įigure 1. Six cases of alopecia areata (AA) in patients treated with infliximab have been reported in the literature ( Table 1). 
This case suggests a complex and contradictory role of TNF-α in the pathogenesis of alopecia We describe a case of alopecia areata universalis associated with infliximab treatment Infliximab, a chimeric monoclonal anti-TNF-alfa agent used to treat autoimmune diseases, has shown a paradoxical side effect Department of Dermatological Sciences, University of Florence, Italy Abstract Allergology and Clinical Immunology, Department of Biomedicine, University of Florence, Florence, ItalyĢ. Letter: Alopecia areata universalis during off-label treatment with Infliximab in a patient with Behçet diseaseĮ Beccastrini 1, D Squatrito 1, G Emmi 1, P Fabbri 2, L Emmi 1 Dermatology Online Journal 16 (9): 15 1.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
